Botanical Carnivory: The Rebels of Flora
- Gülce Tekin

- Mar 4, 2024
- 8 min read
Updated: Mar 11, 2024
Carnivorous plants, a captivating subset of the plant kingdom, have long intrigued scientists and enthusiasts alike with their extraordinary adaptations and predatory habits. These unique botanical specimens have evolved ingenious mechanisms to capture, digest, and absorb nutrients from animal prey, a developmental strategy that sets them apart from their non-carnivorous counterparts. The fascination with carnivorous plants dates back to Charles Darwin's pioneering studies, where he laid the foundation for understanding their evolutionary adaptations. The diverse array of carnivorous plants, boasting over 583 recognized species across 20 genera and five orders, showcases a spectrum of these adaptations—from the iconic Venus flytrap's rapid snap traps to the elegant pitcher plants with pitfall traps and the employment of specialized trap leaves. This article aims to delve into the evolution, diverse adaptations, and ecological significance of carnivorous plants, shedding light on the interplay between flora and fauna in the plant world.
The Origin of Carnivorous Plants
The history of carnivorous plants dates back to the studies of Charles Darwin, highlighting the enduring interest in these plants over the years. In his work Insectivorous Plants (1875), Darwin applied his then-new concept of homology to demonstrate evolutionary and functional convergence among seemingly unrelated plant taxa. Through careful observations and clever experiments, he determined eight genera of plants that directly dissolved animal protein using enzymes similar to pepsin and other proteases, showcasing their carnivorous nature. In addition to these findings, he established that carnivorous plants absorb nutrients directly from the dissolved prey and that captured prey significantly contributes to the plants' development (Ellison & Gotelli, 2009). Instead of the common absorption via the roots pattern of plants, these plants were mostly dependent on their leaves for nutrient absorption.
A century after Darwin's discoveries, many researchers aimed to investigate the evolution of carnivorous traits in plants that are closely related carnivorous species. One study involving sequencing the genomes of carnivorous plants and comparing them to each other as well as non-carnivorous species revealed that the duplication of the entire genome in a common ancestor may have occured approximately 60 million years ago. After the duplication of the whole genome, carnivorous plants tend to repurpose genes associated with nutrient absorption in roots for the absorption of nutrients from digested prey in their leaves. Expression of the genes found in the roots in the leaves of carnivorous plants is an adaptation strategy of these plants, one of the hallmarks of genetic repurposing (Palfalvi et al., 2020). Throughout the years, study of nearly 600 insectivorous plant species allowed researchers to understand the evolution of these plants across different lineages of angiosperms (flowering plants) over time. The recognized insectivorous plant species span a diverse range, distributed among 12 families and five orders of flowering plants (Biswal et al., 2018). This diversity across different angiospermic lineages provides researchers with a rich dataset to analyse and understand the broader evolutionary context of carnivorous plants.
Recent research has conducted genome and transcriptome projects on several carnivorous plants, including Utricularia, Dionaea, Aldrovanda, Drosera, and Cephalotus, and indicate that the gene expression patterns observed in these carnivorous plants are not exclusive to carnivorous traits but rather resemble patterns found in non-carnivorous plants that typically employ defense mechanisms against herbivores and pathogens such as the production of toxic compounds, physical barriers, or other strategies to deter herbivores and pathogens (Hedrich & Fukushima, 2021). In environments with low nitrogen availability, plants face a challenge in obtaining sufficient nitrogen, an essential nutrient for their growth and development in terms of synthesizing proteins and other nitrogen-containing compounds. Studies reveal that the primary motivation for the evolution of carnivorous traits is to supplement the plants' nitrogen budget by capturing animals, especially arthropods and extracting nitrogen from the bodies of their prey, compensating for the lack of nitrogen in the soil (Schulze et al., 1999). Since these plants do not have highly developed root structures to obtain nitrogen from the soil, their dependence on nitrogen is maintained via carnivorous behavior.
What Makes a Plant Carnivorous?
In order for a plant to be considered as carnivorous it must have a clear adaptation to attract, catch, or digest prey as well as having the capability of absorbing nutrients from animals, leading to increased fitness (Böhm & Scherzer, 2021). The use of trap leaves is a common characteristic among carnivorous plants, despite the exceptions such as Triantha occidentalis which uses flypaper-type traps exclusively on its flower stalks (Lin et al., 2021). Carnivorous plants employ these specialized structures, trap leaves, to capture their prey and perform subsequent digestion and absorption. Unlike vertebrate digestive tracts with separate organs for functions like the mouth, stomach, and intestines, trap leaves in carnivorous plants are all-in-one organs. Nevertheless, certain trap types such as Genlisea may exhibit a spatial split of functions within a single leaf where the trapping organs are distinctly separated from the digestive chamber within the same leaf (Freund et al., 2022).
The advantage of carnivorous plants over conventional plant physiology can be analyzed via a cost/benefit analysis, as proposed by Givnish et al. (1984). This concept suggests that a carnivorous lifestyle is only favored when the acquired nutrients from prey surpass the additional costs, such as the energy expended in movement and the production of specialized tissues, organs, and proteins, along with the loss of photosynthetically active tissue. Carnivory becomes viable in areas with limited nutrient availability but abundant light energy and water resources. The evolution of the carnivorous traits involves the neo-functionalization and transformation of existing signaling pathways and innate immunity-related processes to adapt them for a carnivorous lifestyle. So, carnivorous plants repurpose and modify their existing genetic toolkit to thrive in nutrient-limited environments by preying on other organisms (Böhm & Scherzer, 2021).
Hunting Strategies of Carnivorous Plants
The capture organs of carnivorous plants derive from modified leaves and function with two different types of trapping mechanisms. Some carnivorous plant species use immobile (passive) traps, which do not rely on an organ movement for capture purpose (Böhm & Scherzer, 2021). The literature shows that there are five general types of traps in carnivorous plants: adhesive (“flypaper”) traps that have sticky glandular leaves; pitcher (“pitfall”) traps, formed by tubular leaves or, in the case of tank-forming monocots, rosettes of leaves; “snaptraps”, formed by rapidly closing laminar lobes; specialized eel (or “lobster-pot”) traps, formed by narrow, tubular leaves that are internally lined with retrorse hairs; and suction (“bladder”) traps, which are highly modified, sac-like leaves" (Fleischmann et al., 2018).
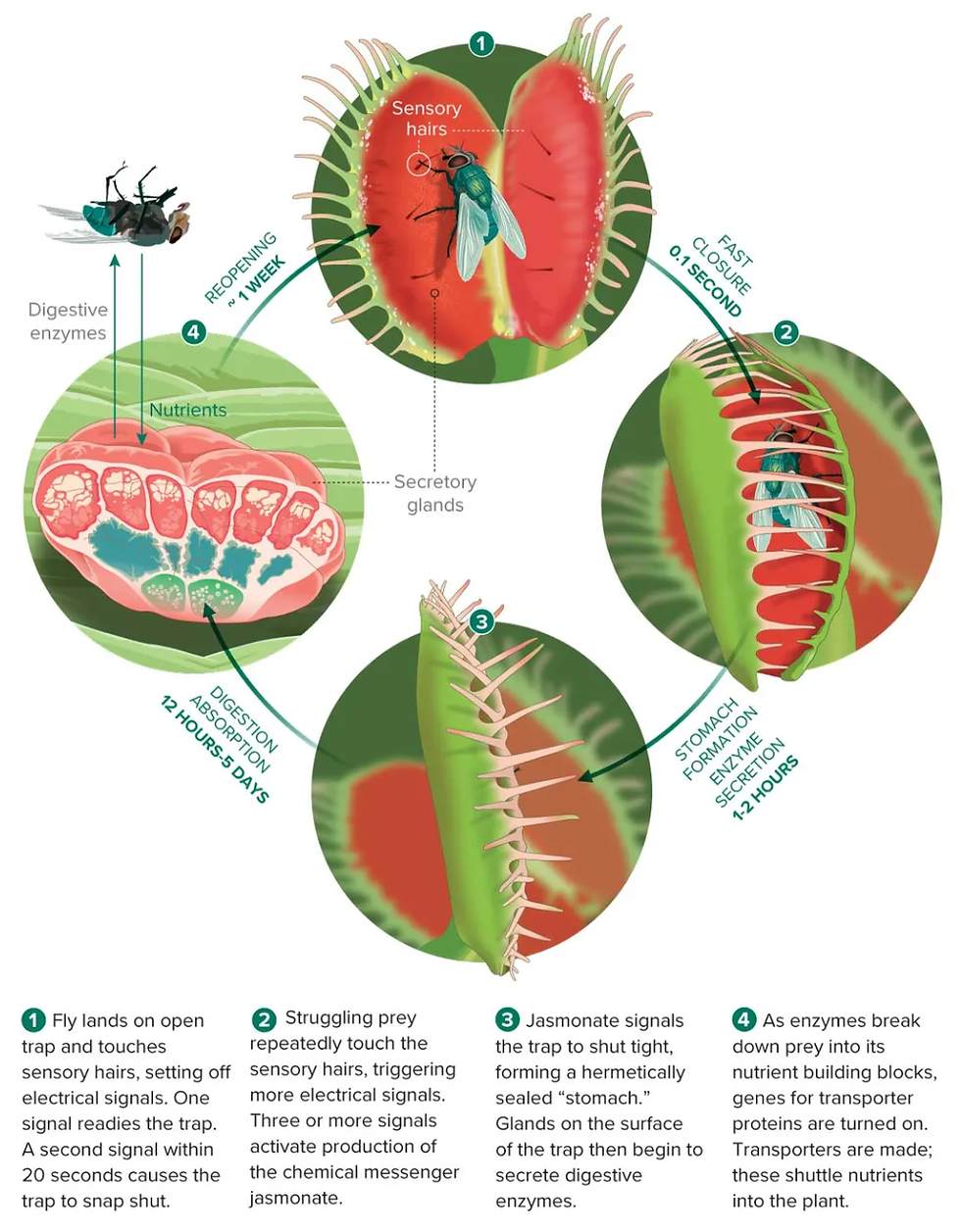
From the inner surface of the carnivorous plant trap, the trigger hairs (consisting of a basal podium and a distal lever connected through a tailed indentation zone) protrude forming a lawn within the digestive glands. The elongated cells surrounding the center of the indentation zone are equipped with mechanosensors that occupy stress when a prey organism, such as an insect searching for food, comes across with the trigger hair. The trigger hairs bend, which serves as the signal that initiates the trapping mechanism of the plant. The mechanosensors found in the sensory zone show specificity and sensitivity towards the prey, ensuing that the trap processes only prey that is not able to escape through the teeth of the closed capture organ. The trap closes only after a trigger hair is stimulated twice, reducing the likelihood of expending energy on unsuccessful capture attempts and providing an energy sufficient strategy of hunting (Hedrich & Fukushima, 2021). The stimulation of mechanosensors initiates the synthesis of jasmonate-type touch hormones which allows the closed trap to hermetically seal, flooding the forming green stomach with lytic enzymes. The prey's outer most shell containing chitin is degraded by hydrolases (chitinases) in the green stomach. Proteins, nucleic acids, glycans, and lipids from the prey are broken down into their respective monomers and macronutrients required for plant growth, including nitrogen, phosphate, sulfate, and minerals like potassium, are released (Hedrich, 2015).
Like many vascular plants, carnivorous plants also have glands that are responsible for prey digestion and nutrient absorption, sharing similarities with other secretory tissues like hydathodes, salt glands, and nectaries. However, these glands of carnivorous plants often have well-differentiated morphologies compared to other epidermal cells (Hedrich & Fukushima, 2021). Digestive glands in carnivorous plants secrete mucilage, ions, and proteins, including digestive enzymes that are known to contribute to the breakdown of prey for subsequent nutrient absorption. The acidity of digestive fluid is considered a hallmark of carnivorous plants and the pH varies among different carnivorous plant genera. The strong acidity has the potential to kill prey organisms while suppressing microbial growth. The acidic pH optimizes the efficiency of digestive enzymes in degrading proteins and other substrates and facilitate nutrient absorption driven by proton gradients. In many carnivorous plant groups such as Sarraceniaceae, the digestive fluid is acidic even in the resting state and becomes more acidic upon prey capture. These species, compared to other pitcher plants, rely more on microbes than their own digestive enzymes, contributing to the lower acidity of the liquid in their pitchers compared to other carnivorous plants (Freund et al., 2022).
The traps of carnivorous plants operate root-type transporters when in contact with trapped animals (Adamec, 2013). After the digestion process, the same or morphologically distinct glands are involved in the absorption of degraded compounds facilitated via membrane transport proteins and endocytosis (Hedrich & Fukushima, 2021). Although partial overlap in functions of glands has been observed, a comprehensive study comparing all relevant genera and different types of glands is necessary to fully understand the complexity and dispel initial assumptions.
Carnivorous plant roots exhibit variability in their functions, both in terms of permanence and seasonal activity. Some species possess deep roots that likely facilitate continuous water uptake throughout the year, while others such as aquatic carnivorous plants have poorly developed roots or even lack roots altogether. The absence of roots does not necessarily imply a lack of function; in certain plants, stems and leaves may assume the functions typically associated with roots. This versatility in root morphology and function reflects the diverse adaptations of carnivorous plants to their specific habitats and nutritional strategies, contributing to their overall ecological success (Adlassnig et al., 2005).
Two important studies focus on carnivorous plants belonging to the Byblis genus, whose trapping mechanism is less explored compared to other carnivorous plant genera. In a study by Poppinga et al. (2022), a series of experiments employing stimulation, kinematics, actuation, and light microscopy were conducted to investigate the stalked trichomes of Byblis gigantea and suggested that the chemonastic movements (chemical-induced movements) of these stalked trichomes may aid Byblis in capturing and digesting its prey. In another study by Li et al., a different Byblis species, B. guehoi, was utilized to test the hypothesis that various leaf trichomes, specifically stalked trichomes and sessile trichomes, serve distinct roles in carnivory. The findings revealed that sessile trichomes are pivotal in nutrient absorption, as they secrete various digestive enzymes and efficiently absorb large protein molecules (Płachno, 2023). This research sheds light on the differentiated functions of different trichome types in Byblis plants during the carnivorous process, enhancing the understanding of their unique predatory strategies.
Conclusion
The world of carnivorous plants acts as a realm of botanical adaptations and ecological strategies. From Charles Darwin's early explorations to contemporary studies, the allure of these green predators lies in their ability to turn the tables on traditional plant-animal relationships by providing nutrients for themselves by digesting insects. The diversity of carnivorous species, boasting various trap mechanisms and root types, underscores the versatility and ingenuity that have evolved across different lineages. Despite the scarcity of quantitative data, the descriptions of roots as "weak" or "strong" hint at the varying roles these below-ground structures play in different species. Some exhibit deep, continuously functional roots, while others have replaced root functions with stems and leaves. These adaptabilities highlight the resourcefulness of carnivorous plants in nutrient-deficient environments. From inconspicuous roots to complex trapping mechanisms, the world of carnivorous plants continues to provide a captivating lens into the interplay between flora and fauna in the natural world!
Bibliographical References
Adamec L. (2013). Foliar mineral nutrient uptake in carnivorous plants: what do we know and what should we know?. Frontiers in plant science, 4, 10. https://doi.org/10.3389/fpls.2013.00010
Adlassnig, W., Peroutka, M., Lambers, H., & Lichtscheidl, I. K. (2005). The Roots of Carnivorous Plants. Plant and Soil, 274(1-2), 127–140. https://doi.org/10.1007/s11104-004-2754-2
Biswal, D. K., Debnath, M., Konhar, R., Yanthan, S., & Tandon, P. (2018). Phylogeny and Biogeography of Carnivorous Plant Family Nepenthaceae With Reference to the Indian Pitcher Plant Nepenthes Khasiana Reveals an Indian Subcontinent Origin of Nepenthes Colonization in South East Asia During the Miocene Epoch. Frontiers in Ecology and Evolution, 6. https://doi.org/10.3389/fevo.2018.00108
Böhm, J., & Scherzer, S. (2021). Signaling and transport processes related to the carnivorous lifestyle of plants living on nutrient-poor soil. Plant physiology, 187(4), 2017–2031. https://doi.org/10.1093/plphys/kiab297
Ellison, A. M., & Gotelli, N. J. (2009). Energetics and the evolution of carnivorous plants—Darwin’s “most wonderful plants in the world.” Journal of Experimental Botany, 60(1), 19–42. https://doi.org/10.1093/jxb/ern179
Fleischmann, A., Schlauer, J., Smith, S. A., & Givnish, T. J. (2018). Evolution of carnivory in angiosperms. In Oxford Scholarship Online. Oxford University Press. https://doi.org/10.1093/oso/9780198779841.003.0003
Freund, M., Graus, D., Fleischmann, A., Gilbert, K. J., Lin, Q., Renner, T., Stigloher, C., Albert, V. A., Hedrich, R., & Fukushima, K. (2022). The digestive systems of carnivorous plants. Plant Physiology, 190(1), 44–59. https://doi.org/10.1093/plphys/kiac232
Givnish T. J. (2015). New evidence on the origin of carnivorous plants. Proceedings of the National Academy of Sciences of the United States of America, 112(1), 10–11. https://doi.org/10.1073/pnas.1422278112
Hedrich, R. (2015). Carnivorous plants. Current Biology, 25(3), R99–R100. https://doi.org/10.1016/j.cub.2014.11.036
Hedrich, R., & Fukushima, K. (2021). On the Origin of Carnivory: Molecular Physiology and Evolution of Plants on an Animal Diet. Annual Review of Plant Biology, 72(1), 133–153. https://doi.org/10.1146/annurev-arplant-080620-010429
Lin, Q., Ané, C., Givnish, T. J., & Graham, S. W. (2021). A new carnivorous plant lineage (Triantha) with a unique sticky-inflorescence trap. Proceedings of the National Academy of Sciences, 118(33), e2022724118. https://doi.org/10.1073/pnas.2022724118
Palfalvi, G., Hackl, T., Terhoeven, N., Shibata, T. F., Nishiyama, T., Ankenbrand, M., Becker, D., Förster, F., Freund, M., Iosip, A., Kreuzer, I., Saul, F., Kamida, C., Fukushima, K., Shigenobu, S., Tamada, Y., Adamec, L., Hoshi, Y., Ueda, K., & Winkelmann, T. (2020). Genomes of the Venus Flytrap and Close Relatives Unveil the Roots of Plant Carnivory. Current Biology, 30(12), 2312-2320.e5. https://doi.org/10.1016/j.cub.2020.04.051
Płachno, B. J. (2023). Carnivorous Plant Biology: From Gene to Traps. International Journal of Molecular Sciences, 24(22), 16179. https://doi.org/10.3390/ijms242216179
Poppinga, S., Knorr, N., Ruppert, S., & Speck, T. (2022). Chemonastic Stalked Glands in the Carnivorous Rainbow Plant Byblis gigantea LINDL. (Byblidaceae, Lamiales). International Journal of Molecular Sciences, 23(19), 11514. https://doi.org/10.3390/ijms231911514
Schulze, W. X., Frommer, W. B., & Ward, J. M. (1999). Transporters for ammonium, amino acids and peptides are expressed in pitchers of the carnivorous plantNepenthes. Plant Journal, 17(6), 637–646. https://doi.org/10.1046/j.1365-313x.1999.00414.x
Visual Sources
Figure 1: Darwin Correspondence Project. University of Cambridge. From:https://www.darwinproject.ac.uk/letters/darwins-works-letters/insectivorous-plants
Figure 2: Schwallier, R., van Wely, V., Baak, M., Vos, R., van Heuven, B. J., Smets, E., van Vugt, R. R., & Gravendeel, B. (2020). Ontogeny and Anatomy of the Dimorphic Pitchers of Nepenthes rafflesiana Jack. Plants, 9(11), 1603. https://doi.org/10.3390/plants9111603
Figure 3: Pain, S. (2022). How Carnivorous Plants Evolved. Smithsonian Magazine. https://www.smithsonianmag.com/science-nature/how-carnivorous-plants-evolved-180979697/
Figure 4: Plants That Bite Back: Keeping Carnivorous Plants. From: https://www.tfhmagazine.com/articles/aquatic-plants/plants-that-bite-back-keeping-carnivorous-plants-full-article
Figure 5: Carnivorous plants with real bite. The Guardian. From: https://www.theguardian.com/lifeandstyle/2018/may/20/carniverous-plants-with-real-bite








What a fascinating read, Gülce! It's amazing how carnivorous plants like the Venus flytrap and pitcher plants have evolved such complex nutrient-gathering strategies—kind of reminds me of how we humans are always finding creative ways to "trap" the best deals. Speaking of which, if anyone here loves discovering unique biological strategies and saving money on coffee runs, I recently compiled a list of the latest Dunkin promo codes that might interest you. Who says science and snacks can’t go hand in hand?
Discover new places with mapquest directions that your go-to tool for detailed maps and step-by-step driving instructions. Enjoy turn-by-turn guidance and real-time traffic updates to avoid delays and find the fastest route.