Biochemistry in Perspective Series: Natural Biochemical Cycles
- Nicole Galetti

- Sep 24, 2023
- 17 min read
Updated: Sep 15, 2024
Foreword
Biological organisms, such as humans and their individual cells, are incredibly complex and diverse systems. Nevertheless, certain unifying characteristics exist in all living things, from the simplest bacterium to the human being. The same types of biomolecules are present and they all use energy to function. These molecules are known as proteins, lipids, glycans, and nucleic acids. From the construction, modification, and interaction of these components, our cells develop and carry out specific functions. Biochemistry draws on a wide range of scientific disciplines to explore and study these molecules, cells, and functions. This has sequentially allowed us to gain a better understanding of the human body at the molecular level, which has led to more effective treatments in medicine. We will explore biochemistry in the context of this series, which examines the role it has played and will continue to play in our daily lives. In a nutshell, all life is the embodiment of biochemistry, and everything a living organism does is an expression of a biochemical process.
This Series is divided into six articles, including:
4. Biochemistry in Perspective Series: Natural Biochemical Cycles
Biochemistry in Perspective Series: Natural Biochemical Cycles
The natural biochemical cycles that take place within a living organism are fundamental processes. These pathways enable the conversion of molecules into energy and metabolic intermediates, which are essential compounds for cellular function and health. All life forms depend on these intricate networks of chemical reactions to function, grow, and maintain themselves. A brief outline of some of the most important biochemical cycles are discussed below.
1. Cellular respiration [Figure 1]:
Glycolysis is the metabolic pathway that converts glucose into pyruvate, and in most organisms, occurs in the cytoplasm of the cell. During the breakdown of glucose, a small amount of adenosine triphosphate (ATP) and nicotinamide adenine dinucleotide (NAD) is generated;
The Krebs cycle (Citric Acid Cycle) is defined as a metabolic pathway that interconnects protein, fat, and protein metabolism. It takes place within the mitochondria, oxidizing pyruvate. Carbon dioxide is released in the process, while ATP, nicotinamide adenine dinucleotide hydride (NADH) and flavin adenine dinucleotide (FADH2) are produced;
Oxidative phosphorylation is the final step in cellular respiration and occurs in the mitochondria of the cell. It is linked to the electron transport chain. During oxidative phosphorylation, energy in the form of ATP is produced;
2. The electron transport chain (ETC) occurs both in cellular respiration and photosynthesis. It is a collection of proteins bound to the inner mitochondrial membrane and organic molecules. Electrons pass through these proteins in a series of energy-releasing reactions;
3. The pentose phosphate pathway is similar to the glycolysis pathway, generating nicotinamide adenine dinucleotide phosphate (NADPH) and pentose sugars in the process (Campbell et al., 2016).

These biochemical cycles are interconnected, which enables efficient utilization of nutrients, essential molecules and energy production. Impairment of these cycles can lead to various metabolic disorders and diseases. Understanding these biochemical pathways is crucial for advancing our knowledge in the field of biochemistry and biotechnology, and developing treatments for medical conditions that are related to these biochemical pathways. Before continuing with the intricacies of each pathway, it is crucial to understand some of the fundamental chemicals and complexes that are present in these cycles. These include NADH, FADH2, and ATP.
Nicotinamide adenine dinucleotide (NAD) is a coenzyme, central to metabolism and found in all living cells. Figure 2 depicts the chemical structure of NAD, the chemical dinucleotide entity consists of two nucleotides bound together by two phosphate groups. The two different nucleotide bases are adenine and nicotinamide. These two nucleotide bases are bonded to two separate ribose groups which are finally connected to the phosphate groups (detailed in Figure 2). NAD is present in two forms; oxidized and reduced (NAD+ or NADH respectively), where it either gains a hydrogen atom or not (Nelson et al., 2008). It was discovered in 1906 by Arthur Haden and William John Young, while conducting an experiment in alcohol fermentation. They realized that adding boiled yeast greatly accelerated the fermentation process when compared to adding yeast that was not boiled (Harden & Young, 1906). The research was continued by Hans von Euler-Chelpin who later extracted and purified the heat-stable factor that was observed by Haden and Young, and identified it as a nucleotide sugar phosphate.
Flavin adenine dinucleotide (FAD), commonly known as quinone, is another type of coenzyme which is presented in various reactions associated with metabolism [Figure 2]. It is a coenzyme that is attached to various proteins and carries out several functions. A flavoprotein is a protein that contains a flavin group, which can be presented in the form of FAD or flavin mononucleotide (FMN). It has four different redox states. FAD interchanges between these states by donating or accepting electrons. In its fully oxidized form, FAD can accept two electrons and two protons to form FADH2 (hydroquinone). The other two forms are semiquinone (FADH) and the super-oxidized form. Flavoproteins were first discovered in 1879 while separating components of cow’s milk. It took a further 50 years for scientists to identify these components as flavoproteins (Abbas & Sibirny, 2011).

Adenosine triphosphate (ATP) is a very important organic molecule that is responsible for many functions inside the living cell. These include; muscle contractions, nerve impulses, and chemical reactions that require the use of energy. When ATP is being used in the body it removes its phosphate groups, forming adenosine diphosphate (ADP) or adenosine monophosphate (AMP). It consists of three components; an adenine base, a ribose sugar and a triphosphate group [Figure 2].
The Components of Cellular Respiration
Glycolysis
Have you ever wondered how yeast ferments barley malt into beer or how your muscles can keep contracting while you are exercising and out of breath? In short, this is due to fermentation and respiration. Glycolysis is the first stage in both of these processes. If we compare what happens to a glucose molecule in human beings as well as a glucose molecule in a bacterium like Lactobacillus acidophilus (a friendly bacterium that turns milk into yogurt, we will notice that the overall metabolism of the glucose molecule would be different, but glycolysis would be its first step in both cases.
Glycolysis is the first step in cellular respiration. Despite this, glycolysis does not require the use of oxygen, which is why many anaerobic organisms, such as the bacterium Lactobacillus acidophilus, have this pathway. It plays a crucial role in energy production and metabolism. In biochemical terms, it is a series of enzymatic reactions that convert one molecule of glucose (a six-carbon sugar molecule, Figure 3) into two molecules of pyruvate (a three-carbon compound, Figure 3). It can be divided into two main phases, the energy-requiring phase and the energy-releasing phase. Both ATP and NADH are produced during the process (Campbell et al., 2016; Nelson et al., 2008). The process is depicted in Figure 3, where the inputs and outputs generated by the process are clearly indicated.

It took almost 100 years for scientists to fully understand the glycolysis pathway. Numerous small experiments were conducted by scientists over time to achieve this. In the early nineteenth century, the wine industry in France sought to investigate why wine sometimes turned distasteful instead of fermenting into alcohol. This prompted French scientist Louis Pasteur in the 1850s to investigate the phenomenon. The results from his experiments initiated the beginning of elucidating the glycolysis pathway. His experiment, known today as the Pasteur effect, shows that oxygen has an inhibiting effect on the yeast fermentation process. He discovered that fermentation takes place by means of living microorganisms (such as yeast) and that glucose consumption by yeast decreases in the presence of oxygen (aerobic conditions), while the opposite was observed in the absence of oxygen (anaerobic conditions).
Eduard Buchner provided insight into several component steps in the glycolysis pathway. This was achieved by his non-cellular fermentation experiments conducted during the 1890s. His experiments demonstrated the enzyme-catalyzed conversion of glucose to ethanol by means of a non-living extract of yeast. This revolutionized the field of biochemistry, allowing scientists to analyze these experiments in a more controlled fashion. A few years later, scientists Arthur Harden and William Young discovered more components of the glycolysis pathway, and the role that ATP played in glycolysis (Singh et al., 2004).
In the 1920s Otto Meyerhoff [Figure 4], linked together several individual reactions, discovered by the many scientists who worked on elucidating the glycolysis pathway. Meyerhoff and his research team made great progress in the glycolysis pathway by extracting different glycolytic enzymes present in the pathway from muscle tissue and simulating these reactions. This finally led to the development of the glycolysis pathway which we know today. It was proposed by Gustav Embden in 1930 [Figure 4], taking into account all the pieces of evidence gathered over the years (Kresge et al., 2005). From then on, the understanding of the pathway has expanded to include further details regarding its regulation and integration with other biochemical pathways. Step-by-step details of the pathway can be seen in Figure 5.

Glycolysis Functions
Energy Production
Energy production is one of the most critical functions of glycolysis. A portion of glucose's potential energy is extracted by glycolysis by breaking it down into pyruvate. The energy produced by glycolysis is used by the cell for various other cellular processes and functions. Though glycolysis itself is not the most efficient pathway for ATP generation, it is a fast and readily available source of energy, making it essential, especially in situations where energy demands are immediate, such as muscle contraction during physical activity.
Anaerobic respiration
In anaerobic conditions where oxygen availability is limited or absent, glycolysis is particularly important. Although glucose is completely oxidized through subsequent biochemical processes, generating copious amounts of ATP, it is still generated through glycolysis without the need for oxygen. This is especially vital for microorganisms that thrive in anaerobic environments and for tissues like muscle cells during strenuous exercise when the oxygen supply may be insufficient to meet energy demands.
Glucose homeostasis
Glycolysis plays a crucial role in glucose homeostasis. Glucose homeostasis is the balance of insulin and glucagon to maintain blood glucose levels. According to the body's needs, it can either provide glucose or dispose of it. At low blood glucose levels, gluconeogenesis (the reverse of glycolysis) is initiated to produce glucose from non-carbohydrate sources such as lactate and pyruvate. When blood glucose levels are high, excess glucose is stored as glycogen. When there is a need for energy again, glycogen is broken down to glucose for ATP production.
Interplay with Several Biochemical Pathways
The glycolysis pathway is not isolated from other metabolic pathways, but rather interacts with them all. Its intermediates are crucial precursors for other biosynthetic processes, such as the pentose phosphate pathway, essential for lipid metabolism. These connections emphasize the central role of glycolysis in overall metabolism.

Understanding the intricacies of glycolysis is essential for comprehending the fundamentals of cellular metabolism, and has profound implications for fields such as medicine, biochemistry, and biotechnology. As such, glycolysis continues to be a focus of research, offering insight into basic biology as well as potential therapeutic interventions.
Significance of Glycolysis in Biology and Medicine
Diabetes
Insulin is released by the pancreas in response to rising glucose levels in the bloodstream. Insulin then helps the blood sugar enter the cells, so that it can be broken down through glycolysis and be used for energy. This, in turn, lowers blood sugar levels. Persons who suffer from diabetes have either low or no insulin levels, which can result in hyperglycemia. Hyperglycemia occurs when blood sugar levels are too high, and glucose cannot be broken down to pyruvate. Gluconeogenesis (the process of making glucose in the body or the reverse of glycolysis) further contributes to hyperglycemia in diabetes (Scaroni et al., 2017). The role of glycolysis is extremely important to maintain blood sugar levels within the body and prevent diabetes.
Genetic Diseases
Glycolytic mutations affecting glucose metabolism are generally rare; most mutations result in early cell death. There are, however, some mutations such as glycogen storage diseases and inborn errors of carbohydrate metabolism that can be observed, with pyruvate kinase deficiency being one notable example. Pyruvate kinase is the enzyme that is involved in the last step of glycolysis, producing pyruvate and ATP. Pyruvate kinase deficiency is a condition in which red blood cells break down faster than they should (Zanella et al., 2005), leading to anemia (insufficient amount of red blood cells).
Cancer
Malignant tumor cells perform glycolysis ten times faster when compared to noncancerous tissue cells. Specific glycolytic enzymes are over-expressed in some tumor cells, resulting in faster glycolysis. This is known as the Warburg effect, described by Otto Warburg in 1930. The Warburg hypothesis claims that cancer is mainly caused by dysfunctional mitochondria, rather than the uncontrolled growth of cells. Basically, cancer cells produce energy through a less efficient process of anaerobic glycolysis consisting of a high level of glucose uptake followed by lactic acid fermentation taking place in the cytosol. Aerobic glycolysis takes place in the mitochondria generating a lot more ATP with a lot less glucose when compared to anaerobic glycolysis, as it can be seen in the diagram depicted in Figure 6 (Alfarouk et al., 2014).

Krebs Cycle
There are various names for the Krebs cycle, in particular the citric acid cycle and the tricarboxylic acid cycle. It is often described as the hub of cellular respiration. It is named after Sir Hans Adolf Krebs. Adolf Krebs was a German-born biochemist and pioneer in cellular respiration research. He discovered that various metabolic processes within the cell are interconnected through the citric acid cycle. During this biochemical process, energy is generated, vital biomolecular intermediates are produced, and important coenzymes and cofactors are synthesized and are later used in other biochemical cycles. (Leigh, 2009).
The Krebs cycle is the second phase of cellular respiration. It occurs inside the mitochondria in eukaryotic cells. It follows a series of reactions that oxidize acetyl coenzyme A (acetyl-CoA), a two-carbon molecule, connected to coenzyme A. In the first step of the cycle, acetyl-CoA combines with a four-carbon acceptor molecule, oxaloacetate, to form citrate. The subsequent reactions are catalyzed by various enzymes that regulate the citric acid cycle and form carbon dioxide, ATP and reducing agents, such as NADH and FADH2, in the process. These reducing agents carry high-energy electrons to the electron transport chain, which ultimately leads to creating more ATP. The entire process can be seen in the diagram below (Campbell et al., 2016) [Figure 7].

Important Functions of the Krebs Cycle
Energy Production
Energy is directly harvested from acetyl-CoA by the Krebs cycle. Acetyl-CoA, derived from various sources like glucose, fatty acids, and several amino acids, enters the citric acid cycle and undergoes a series of reactions, releasing cofactors that carry electrons to the electron transport chain, where the creation of a proton gradient is used to synthesize ATP. The energy produced is important in muscle contraction, as well as in maintaining and repairing cells.
Carbon Skeleton for Biosynthesis
In addition to producing energy, the Krebs cycle provides carbon skeletons for the biosynthesis of essential molecules. As one could expect, several biomolecules (produced as intermediates in the cycle) such as oxaloacetate and α-ketoglutarate, are used as building blocks to synthesize various other amino acids [Figure 8], nucleotides and biomolecules. In this way, the Krebs cycle plays a crucial role in maintaining the integrity and growth of the cell.
Regulation of Metabolism
There are several pathways connected to the Krebs cycle, including glycolysis, fatty acid oxidation, and the pentose phosphate pathway. It acts as a metabolic switch, responding to the cell's energy needs and regulating the flow of carbon and electrons through various pathways. In this way, available resources are efficiently utilized, waste is prevented, and metabolic flexibility is increased.
Amplification of Electron Transport
A crucial role of the Krebs cycle is its contribution to the ETC. The NADH and FADH2 coenzymes produced during the citric acid cycle carry high-intensity electrons to the ETC, ultimately driving the production of ATP synthesis. As a result of this amplification of electrons during the Krebs cycle, ATP production is enhanced and the cycle becomes indispensable in the oxidative phosphorylation process.

Pyruvate Oxidation
The next step to be looked at is pyruvate oxidation, which is a fundamental step in cellular respiration but is often undervalued. It plays a pivotal role in extracting energy from glucose and other carbon sources, as it bridges the gap between glycolysis and the citric acid cycle. The journey of pyruvate oxidation starts with glycolysis. As discussed previously, one molecule of glucose is converted to two molecules of pyruvate during glycolysis. Pyruvate is then transformed to acetyl-CoA. This occurs inside the mitochondria, where specialized transporter molecules facilitate the movement of pyruvate into the mitochondria (Campbell et al., 2016).
Inside the mitochondria, pyruvate encounters the pyruvate dehydrogenase enzyme complex, where pyruvate is oxidized leading to the removal of carbon dioxide and the production of NADH. The rest of the pyruvate molecule binds with coenzyme A (CoA), forming acetyl-CoA which subsequently enters the citric acid cycle. The steps that follow ultimately lead to the production of ATP (Ahmad et al., 2018) [Figure 9].

Oxidative Phosphorylation
Oxidative phosphorylation is the final step of cellular respiration. It takes place in the inner membrane of the mitochondria, where a series of protein complexes and electron carriers work together to generate ATP. The process begins with the electron transport chain. The ETC consists of a series of complexes (I to IV) embedded into the intermembrane of the mitochondria indicated in Figure 10. During this process, coenzymes NADH and FADH2 carry high-energy electrons through these complexes, releasing energy in the process. As electrons move through the ETC, hydrogen protons are pumped from the inner membrane to the intermembrane space generating a proton gradient, otherwise known as the proton motive force. Here potential energy is stored. The enzyme complex ATP synthase harvests the potential energy in the inner membrane to synthesize ATP (Ahmad et al., 2018).

The importance of energy production cannot be emphasized enough. Oxidative phosphorylation is;
the primary method by which cells generate ATP, which fuels virtually every cellular process in our bodies, from muscle contraction to DNA replication,
it is flexible and can be adapted to meet varying energy demands; when energy demands are high, ATP production can increase, while when energy demands are low, it can slow down,
oxidative phosphorylation is equally important in maintaining mitochondrial health, as dysfunctional oxidative phosphorylation can lead to premature aging and various health issues. In living cells, oxidative phosphorylation is believed to generate 90% of the ATP required. A single NADH molecule can produce three ATP molecules and a single FADH2 molecule can produce up to two ATP molecules, this efficiency ensures that the maximum amount of energy is harnessed from nutrients and biomolecules (Ahmad et al., 2018).
The process of oxidative phosphorylation holds importance beyond cellular biology and has interdisciplinary significance in medicine, evolution, and bioenergetics. Dysfunctions in oxidative phosphorylation have been known to be the primary cause in certain mitochondrial and neurodegenerative diseases, i.e. Parkinson’s disease, Alzheimer’s disease, amyotrophic lateral sclerosis, and less commonly, multisystem atrophy and progressive supranuclear palsy. Understanding oxidative phosphorylation is key to developing treatments and therapies for these diseases.

Oxygen and Its Importance in Cellular Respiration
Oxygen is vital for our survival, as it is for nearly all organisms. It is known that not getting enough oxygen could lead to dizziness, blackouts, and even death. This is the reason why our cells require oxygen, specifically during the process of oxidative phosphorylation. It comes into play at the end of the electron transport chain as seen in Figure 10. Here oxygen accepts electrons and protons to form water. If oxygen is absent (for example when someone stops breathing because there is not enough oxygen available) and therefore unable to accept electrons, the electron transport chain would stop generating ATP, and our cells would eventually die due to the lack of ATP.
Pentose Phosphate Pathway
The pentose phosphate pathway (PPP), sometimes referred to as the hexose monophosphate shunt, is a complex metabolic pathway that operates in the cytoplasm of cells. The process stems from glycolysis and performs numerous vital functions, making it one of the cornerstones of cellular metabolism. These functions include synthesizing nicotinamide adenine dinucleotide phosphate (NADPH), synthesizing pentose sugars, reducing glutathione (GSH), and detoxification. NADPH is a key coenzyme that plays a vital role in metabolism including antioxidant defense, biosynthesis of lipids and amino acids, and protection against toxicity of reactive oxygen species. Pentose sugars are five-carbon sugars that are essential for the synthesis of nucleotides (the building blocks of RNA and DNA). The PPP helps to maintain the reduced form of glutathione, which is a powerful antioxidant. GSH helps protect cells from oxidative stress by neutralizing harmful free radicals. Finally, the PPP helps to detoxify harmful chemicals that may be present in the cell (Stincone et al., 2015).
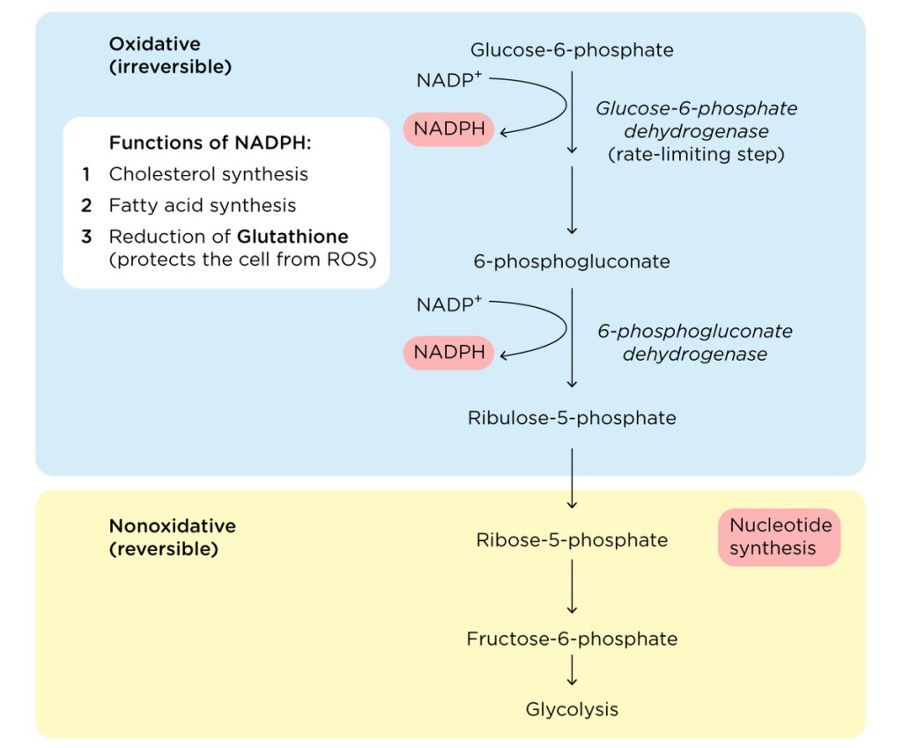
Significance in Biology and Medicine
Cancer metabolism
An altered metabolism is one of the hallmarks of cancer cells. It is common for cancer cells to become more dependent on the PPP in order to meet high energy and biosynthesis demands. Identifying the role of PPPs in cancer metabolism may lead to the development of new cancer treatments (TeSlaa et al., 2023).
Nucleotide synthesis
The PPP contributes towards synthesizing pentose ribose sugars, which are important in nucleotide synthesis. This in turn helps DNA and RNA repair and replicate, processes which are vital for cell division and transferring genetic information.
Antioxidant defense
NADPH generated by the PPP is a key component of the cellular antioxidant system [Figure 13]. It helps protect cells from damage caused by oxidative stress, a factor implicated in aging and various diseases, including cancer.
Neurological diseases
It has been found that disruptions in the PPP have been linked to neurodegenerative diseases, such as Alzheimer's and Parkinson's. It may be possible to gain insights into disease mechanisms and potential therapeutic targets from this area of research (TeSlaa et al., 2023).

Conclusion
Cellular respiration and metabolism are fundamental processes that sustain life. As the cornerstone of energy production, cellular respiration converts nutrients into ATP, the universal currency of energy. It is through a series of interconnected reactions that cells extract energy from glucose and other organic molecules, providing the necessary fuel for various cellular activities.
The intricate network of biochemical pathways within living organisms serves as the foundation of life itself. The metabolic pathways encompass a wide variety of metabolic processes, each with its own set of enzymes, substrates, and intermediates, which work harmoniously to maintain cellular homeostasis. These pathways show how biochemical systems are remarkable in their adaptability and efficiency, from glycolysis to Krebs cycle to pentose phosphate pathway.
In addition to being fundamental to biochemistry and molecular biology, understanding these pathways has profound implications for medicine, agriculture, and biotechnology. The evolutionary history of these biochemical cycles has shed light on the early origins of life and the development of eukaryotic cells. Furthermore, scientists have used and keep using the basic principles of energy conversion in these pathways to understand and develop new clean energy solutions.
The exploration of biochemical pathways is ultimately a journey of discovery, revealing the elegance and complexity of life's inner workings. In this fascinating field of biology, we are bound to discover new insights and innovations that will drive science forward and improve our understanding of nature.
Bibliographical References
Abbas, C. A., & Sibirny, A. A. (2011). Genetic control of biosynthesis and transport of riboflavin and flavin nucleotides and construction of robust biotechnological producers. Microbiology and molecular biology reviews, 75(2), 321–360.
Ahmad, M., Wolberg, A., & Kahwaji, C. I. (2022). Biochemistry, electron transport chain. In StatPearls. StatPearls Publishing.
Alfarouk, K. O., Verduzco, D., Rauch, C., Muddathir, A. K., Adil, H. B., Elhassan, G. O., Ibrahim, M. E., Orozco, J. D. P., Cardone, R. A., & Reshkin, S. J. (2014). Glycolysis, tumor metabolism, cancer growth and dissemination. A new pH-based etiopathogenic perspective and therapeutic approach to an old cancer question. Oncoscience, 1(12), 777.
Campbell, M. K., Farrell, S. O., & McDougal, O. M. (2016). Biochemistry. Cengage Learning.
Harden, A., & Young, W. J. (1906). The alcoholic ferment of yeast-juice. Part II.—The coferment of yeast-juice. Proceedings of the Royal Society of London. Series B, Containing Papers of a Biological Character, 78(526), 369–375.
Kresge, N., Simoni, R. D., & Hill, R. L. (2005). Otto Fritz Meyerhof and the elucidation of the glycolytic pathway. Journal of Biological Chemistry, 280(4), 3.
Leigh, F. (2009). Sir Hans Adolf Krebs (1900–81), pioneer of modern medicine, architect of intermediary metabolism. Journal of Medical Biography, 17(3), 149–154.
Nelson, D. L., Lehninger, A. L., & Cox, M. M. (2008). Lehninger principles of biochemistry. Macmillan.
Reid, D. M., Barber, R. C., Thorpe, R. J., Sun, J., Zhou, Z., & Phillips, N. R. (2022). Mitochondrial DNA oxidative mutations are elevated in Mexican American women potentially implicating Alzheimer’s disease. npj Aging, 8(1), 2. https://doi.org/10.1038/s41514-022-00082-1
Scaroni, C., Zilio, M., Foti, M., & Boscaro, M. (2017). Glucose metabolism abnormalities in Cushing syndrome: from molecular basis to clinical management. Endocrine reviews, 38(3), 189–219.
Singh, P., Batra, H., & Naithani, M. (2004). History of biochemistry. Bulletin of the Indian Institute of History of Medicine (Hyderabad), 34(1), 75–86.
Stincone, A., Prigione, A., Cramer, T., Wamelink, M. M., Campbell, K., Cheung, E., Olin-Sandoval, V., Grüning, N. M., Krüger, A., Tauqeer Alam, M., Keller, M. A., Breitenbach, M., Brindle, K. M., Rabinowitz, J. D., & Ralser, M. (2015). The return of metabolism: biochemistry and physiology of the pentose phosphate pathway. Biological Reviews of the Cambridge Philosophical Society, 90(3), 927–963. https://doi.org/10.1111/brv.12140
TeSlaa, T., Ralser, M., Fan, J., & Rabinowitz, J. D. (2023). The pentose phosphate pathway in health and disease. Nature Metabolism, 5(8), 1275–1289.
Zanella, A., Fermo, E., Bianchi, P., & Valentini, G. (2005). Red cell pyruvate kinase deficiency: molecular and clinical aspects. British Journal of Haematology, 130(1), 11–25.
Visual Sources
Cover Image: Canva. (n.d.). Retrieved September 18, 2023 from: https://www.canva.com/photos/MADq3y-1E3Q-f021-4125/
Figure 1: Khan Academy. (2007). Retrieved September 18, 2023 from: https://www.khanacademy.org/science/biology/cellular-respiration-and-fermentation/variations-on-cellular-respiration/a/connections-between-cellular-respiration-and-other-pathways
Figure 2: Wikipedia. (2022, 2015 & 2007). Retrieved September 18, 2023 from: https://en.wikipedia.org/wiki/File:FAD.png; https://en.wikipedia.org/wiki/Adenosine_triphosphate#/media/File:Adenosintriphosphat_protoniert.svg; https://en.wikipedia.org/wiki/Nicotinamide_adenine_dinucleotide#/media/File:NAD+.svg
Figure 3: Embibe. (n.d.). Retrieved September 18, 2023 from: https://www.embibe.com/exams/glycolysis/
Figure 4: Nobelprize & Wikipedia. (1922 & 2022). Retrieved September 18, 2023 from: https://www.nobelprize.org/prizes/medicine/1922/meyerhof/biographical/
https://en.wikipedia.org/wiki/Gustav_Embden#/media/File:GustavEmbden_2_(cropped).jpg
Figure 5: Wikipedia. (2015). Retrieved September 17, 2023 from: https://en.wikipedia.org/wiki/File:Glycolysis_metabolic_pathway_3_annotated.svg
Figure 7: Istock Photos. (2020). Retrieved September 18, 2023 from: https://www.istockphoto.com/fr/vectoriel/diagramme-de-cycle-dacide-citrique-sch%C3%A9ma-mol%C3%A9culaire-dillustration-vectorielle-gm1207501078-348666315
Figure 10: Microbe Notes. (n.d.). Retrieved September 18, 2023 from: https://microbenotes.com/electron-transport-chain/
Figure 11: Reid, D. M., Barber, R. C., Thorpe, R. J., Sun, J., Zhou, Z., & Phillips, N. R. (2022). Mitochondrial DNA oxidative mutations are elevated in Mexican American women potentially implicating Alzheimer’s disease. npj Aging, 8(1), 2. https://doi.org/10.1038/s41514-022-00082-1
Figure 12: Jack Westin. (n.d.). Retrieved September 18, 2023 from: https://jackwestin.com/resources/mcat-content/glycolysis-gluconeogenesis-and-the-pentose-phosphate-pathway/pentose-phosphate-pathway







Comments